Three Industries With High Quality Assurance Standards


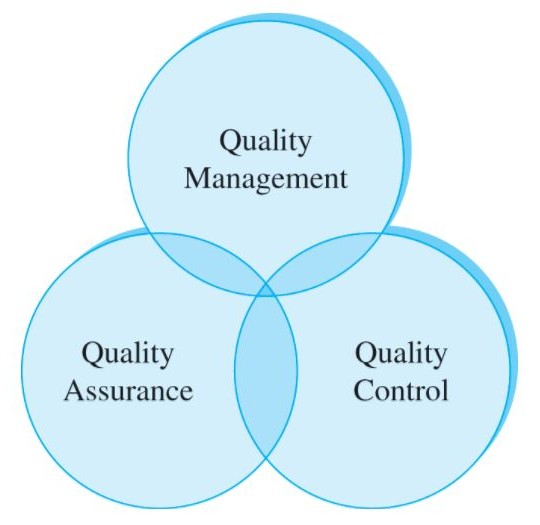
- Quality Assurance (QA): The high-level proof submitted to the public, and—more importantly—regulators that your products are of sufficient quality.
- Quality Management System (QMS): A series of processes that delivers quality assurance by implementing quality control.
- Quality Control (QC): The “boots-on-the-ground” act of rooting out and eliminating problems that might hinder quality production.
Pharmaceutical Industry

- Public Safety: Medications can change someone’s life for the better or cause serious harm. Quality controls to protect public safety include strict attention to document control, employee training, laboratory OOS, corrective and preventive action (CAPA), internal audits, and managerial review.
- Professional and Consumer Confidence: Edelman’s 2019 Trust Barometer rated the public trust in the pharma industry at 57%, worse than any other healthcare vertical. Robust attention to quality assurance is necessary now more than ever to regain consumer and professional confidence.
- Production Efficiency: Production efficiency allows safe products to be produced at a lower cost, increasing profits and fostering affordability severely lacking in the industry. Companies can improve production efficiency through automated data collection and holistic data integration.
Medical Devices Industry

- Physical Devices: Medical devices that live at or come from a hospital, like MRI machines, pacemakers, and defibrillators.
- IoT Devices: Short for “internet of things,” IoTs are a class of physical device that perform functions via the internet. Examples include blood glucose monitors and dissolving EEG monitors that transmit data by wireless signal or into the cloud.
- SaMD: SaMD--“Software as a Medical Device”--refers to software that performs the function of a medical device, regardless of the device it is in (i.e. smartphones, smart watches, tablets, etc.) Examples include software that uses the microphone or accelerometer in a smartphone to record breathing data or tremors.
- Corrective and Preventative Actions (CAPA): Procedures to correct quality management failures.
- Complaint Procedures: How to receive, document, and respond to customer complaints, including tracking and close-out procedures.
- Reporting Procedures: Including written procedures for reporting data and key descriptions in compliant form.
- Nonconforming Product Procedures: Nonconforming products and materials are commonplace and acceptable if appropriate controls are applied.
Aerospace Industry
The aerospace industry sits at the intersection of multiple regulatory purviews, resulting in one of the heaviest compliance burdens of any industry. Millions of consumers who depend on commercial aviation and medivac helicopters are counting on the industry’s compliance and safety. Regulatory agencies with oversight over the aerospace industry include the Federal Aviation Authority (FAA), the Department of Defence (DOD), the National Aeronautics and Space Administration (NASA), and the Environmental Protection Agency (EPA)--and that’s just in the US. The key quality assurance control that applies to aerospace manufacturing is AS9100, also known as AS9100D due to Amendment D. This standard was created by the International Organization for Standardization to adapt quality assurance control standard ISO 9001 to the specific needs of the aerospace industry. Dickson states that the standard is quite comprehensive, noting that it covers company policies, procedures, and documentation. Not only do aerospace manufacturers have to comply with AS9100D, but so too do their suppliers and subcontractors. International agencies, like the EU, often adopt versions of the standard that are basically identical. AS9100 is also divided into three standards:- AS9100: Aerospace design, development, and manufacture.
- AS9110: Aircraft maintenance organizations.
- AS9120: Distributors of hardware, electronics, and other materials.
- Development and Design: Standards for new products.
- Purchasing: Selection of vendors, purchasing procedures, validation and verification.
- Traceability: Being able to trace processes to the accountable party.
- Risk Management: Reducing QC risks where possible, accepting and managing risks where not.
×
![]()