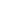Interfacing of Arduino with GLCD

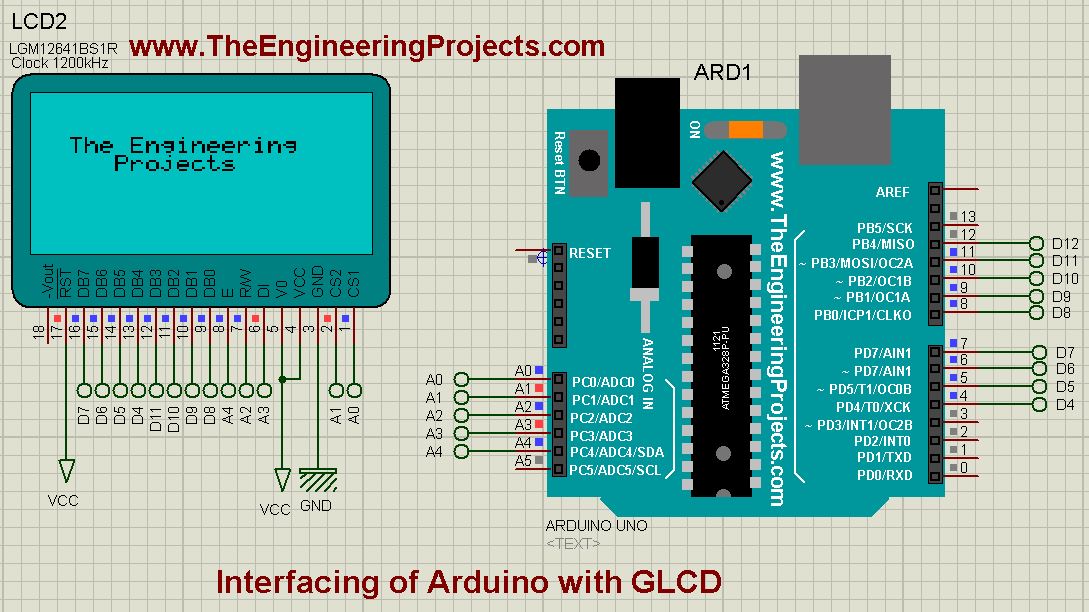
Interfacing of Arduino with GLCD
- First of all, you can download the Proteus Simulation and Arduino Code for Interfacing of Arduino with GLCD, by clicking the below button:
- Now let's design it so that you can understand how this is working.
- So, first of all design a Proteus Simulation for Interfacing of Arduino with GLCD, as shown in below figure:
- Now upload the below Arduino code in your Arduino Software and Get your Arduino Hex File, which we are gonna upload in our Proteus Arduino.
- Here's the Arduino Code for Interfacing of Arduino with GLCD:
- You also have to install the GLCD Library for Arduino, I have added this library in the above package so when you download it first of all install this library in Arduino Software.
#include <glcd.h>
#include "fonts/allFonts.h"
#include "bitmaps/allBitmaps.h"
Image_t icon;
gText textArea;
gText textAreaArray[3];
gText countdownArea = gText(GLCD.CenterX, GLCD.CenterY, 1, 1, Arial_14);
unsigned long startMillis;
unsigned int loops = 0;
unsigned int iter = 0;
int theDelay = 20;
void setup()
{
GLCD.Init();
if(GLCD.Height >= 64)
icon = ArduinoIcon64x64;
else
icon = ArduinoIcon64x32;
GLCD.ClearScreen();
GLCD.SelectFont(System5x7, BLACK);
GLCD.CursorTo(2, 2);
GLCD.print("The Engineering");
GLCD.CursorTo(5, 3);
GLCD.print("Projects");
}
void loop()
{
}
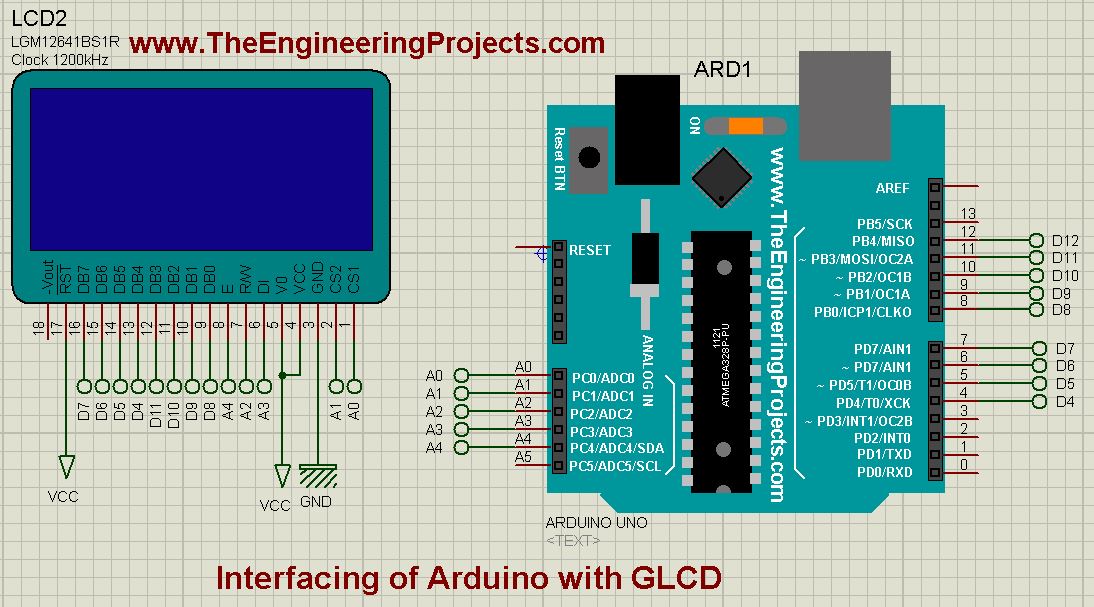
- So, now if everything goes fine then when you run your Proteus Simulation of Arduino with GLCD, you will get results as shown in below figure:

- So, what we have done is we just printed our blog name on the GLCD using Arduino.
- Now, in the package you download I have also added another example which when you upload will give you a demo of GLCD.
- Here's the results of the second example, I have added some screenshots:
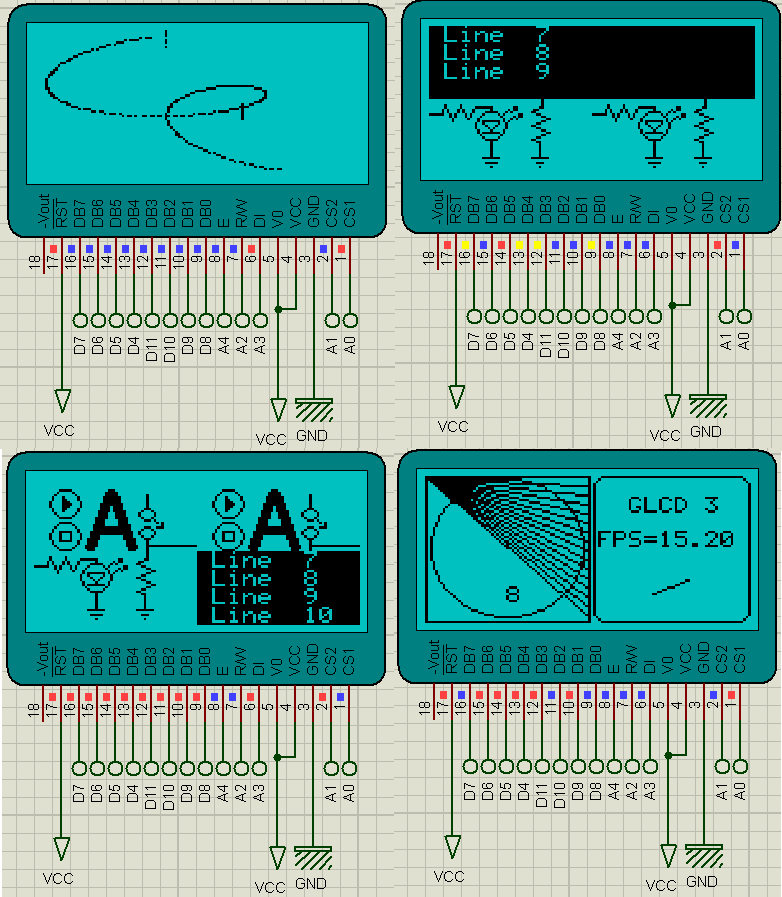
- So, that's how you can interface Arduino with GLCD and can design anything you want.
- It's really very easy but quite lengthy, I must tell.
- I have designed this video which will help you in better understanding:
×
![]()